 
(c) C 3 H 8 ( g ) + 5 O 2 ( g ) ⇌ 3 CO 2 ( g ) + 4 H 2 O ( g ) x 5 x −3 x −4 x C 3 H 8 ( g ) + 5 O 2 ( g ) ⇌ 3 CO 2 ( g ) + 4 H 2 O ( g ) x 5 x −3 x −4 xĬomplete the changes in concentrations for each of the following reactions: (b) I 2 ( a q ) + I − ( a q ) ⇌ I 3 − ( a q ) − x − x x I 2 ( a q ) + I − ( a q ) ⇌ I 3 − ( a q ) − x − x x 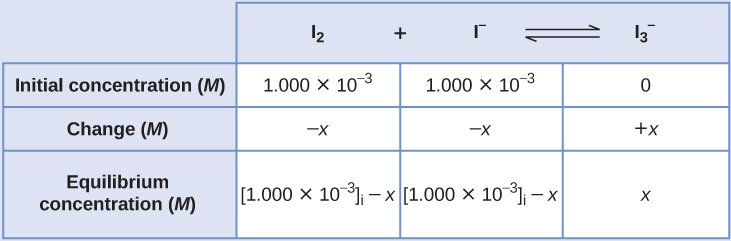
(a) C 2 H 2 ( g ) + 2 Br 2 ( g ) ⇌ C 2 H 2 Br 4 ( g ) x 2 x − x C 2 H 2 ( g ) + 2 Br 2 ( g ) ⇌ C 2 H 2 Br 4 ( g ) x 2 x − x (b) I 2 ( a q ) + I − ( a q ) ⇌ I 3 − ( a q ) _ _ x I 2 ( a q ) + I − ( a q ) ⇌ I 3 − ( a q ) _ _ x These terms are derived from the stoichiometry of the reaction, as illustrated by decomposition of ammonia:ĭetermining Relative Changes in Concentrationĭerive the missing terms representing concentration changes for each of the following reactions. Many of the useful equilibrium calculations that will be demonstrated here require terms representing changes in reactant and product concentrations. Knowledge of the quantitative aspects of these equilibria is required to compute a dosage amount that will solicit the desired therapeutic effect. 
After a drug is ingested or injected, it is typically involved in several chemical equilibria that affect its ultimate concentration in the body system of interest. These types of computations are essential to many areas of science and technology-for example, in the formulation and dosing of pharmaceutical products. Having covered the essential concepts of chemical equilibria in the preceding sections of this chapter, this final section will demonstrate the more practical aspect of using these concepts and appropriate mathematical strategies to perform various equilibrium calculations.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
